When developing sensification systems for personal healthcare, monitoring, and diagnoses, designers need to consider many factors that contribute to the integrity of a sensed signal and to the ability of a system to operate reliably, regardless of the environment.
The sensor itself needs to be non-invasive and comfortable to wear, while being accurate and stable across a range of temperature and humidity conditions, particularly as the user gets more active. At the same time, cables, terminals and connectors need to be able to reliably transport low-level analog signals from the sensor to be conditioned, digitized and sent to the main processor for analysis. Much depends upon the integrity of the data as decisions will be made based on the data analysis.
To achieve the highest level of integrity, designers need to take a holistic approach to system design, not just the sensing circuit itself. The device must be highly immune to electromagnetic interference (EMI), structurally robust, and as moisture proof as possible so it can be cleaned regularly without concern about moisture ingress.
Once the core components of the sensification system have been determined and their performance requirements defined, it’s critical to look at component vendors and suppliers. These play a critical role and can be evaluated and differentiated based on their proven ability to deliver high-quality components, globally with knowledgeable technical support and fast response times. Ideally, one qualified supplier could provide all, or most, of the components to ensure consistent quality and delivery, and simplify inventory management.
Often it pays to connect early in the design process with such a supplier and partner with them before making the final design choices, as their experience across multiple applications and customers can help refine and optimize the system and avoid errors up front.
Sensification components
Of the many components required, sensors are ones that initiate the chain of events. They measure heart rate, blood oxygen levels and flow rate, temperature, humidity, pressure and patient weight distribution, and detect anomalies, such as bubbles in intravenous fluid feed lines. While sensors vary in what they may be detecting, and assuming they are qualified to operate in medical environments, the performance requirements are similar across the board: simplicity, accuracy, reliability, and ruggedness, all while being minimally invasive.
An example of a sensor that meets these requirements is the AD-101 air bubble detector (Figure 1). Air bubble detection is important in applications such as infusion pumps, hemodialysis, and blood flow monitoring, as an undetected bubble in the bloodstream can cause a heart attack, stroke, or respiratory failure.
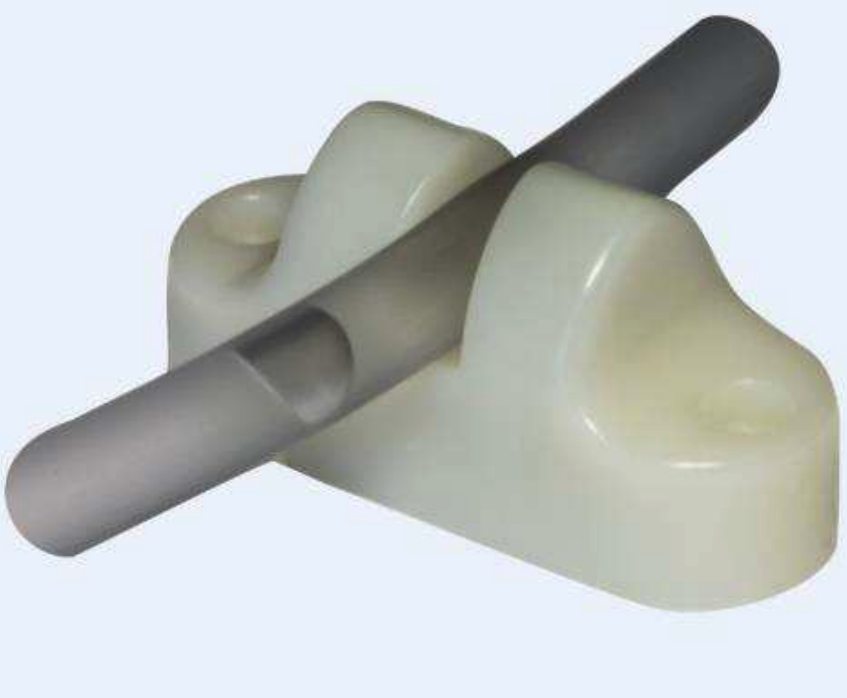
Ideally, detection would be done non-invasively to avoid sterility and fluid compatibility concerns, and without interfering with the intravenous flow itself. The AD-101 meets these requirements using ultrasonic technology and its accuracy allows it to detect a bubble as small as 70% of the inner tube diameter.
As it uses ultrasound, it is inherently immune to EMI and radio-frequency interference (RFI), and to simplify design, it has all required electronics within the package (Figure 2). The four-wire interface comprises the supply (5 to 24 VDC), ground (GND), and two binary outputs: signal output (bubble = logic “1”, liquid = logic “0”), and a self-test output (test OK = logic high, test fault = logic 0).
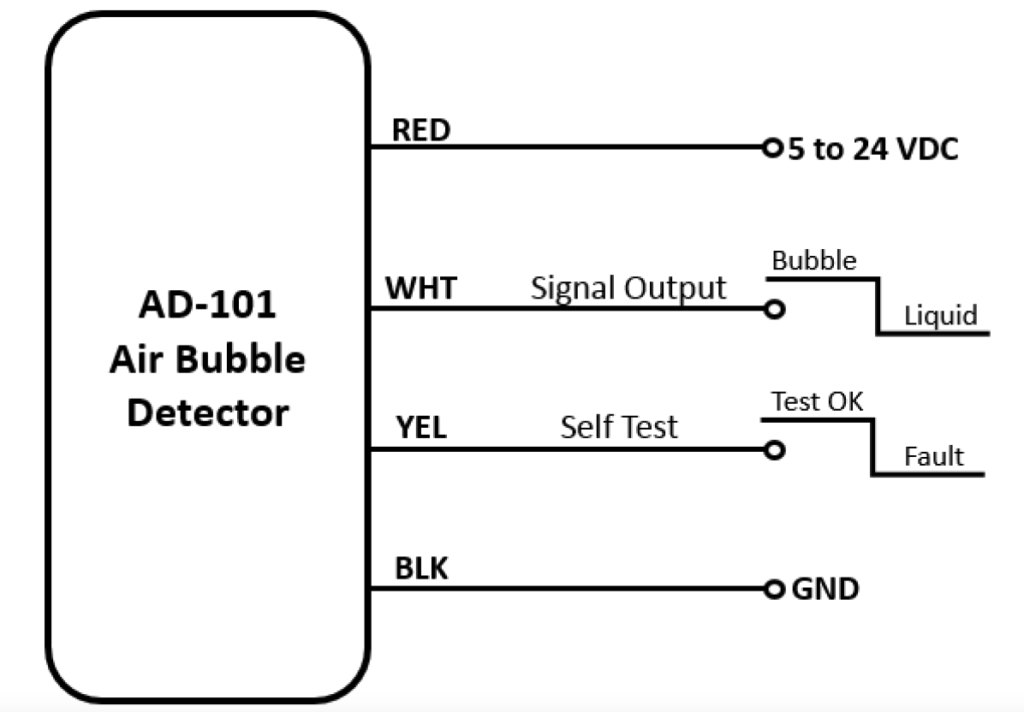
For any sensor measuring a critical parameter, built-in self-test is essential to ensuring sensor reliability and data integrity, but it can add circuit complexity. Having both the sensor electronics and self-test in one sealed package greatly simplifies sensor implementation.
The cable comprises the four 28 AWG conductors and measures 350 mm long, with non-integral jacketing that conforms to UL 2725 such that it tolerates temperatures up to 80˚C. The AD-101 itself operates across the range of 0˚ to 40˚C and performance wise it has a fast response time of just 0.22 ms, which can be critical for fast-moving fluids.
The sensor accommodates standard tube sizes of 4 mm and 6 mm but can be customized to tubing outer diameters of between 3 mm and 10 mm. This results in variations of the tube holder assembly dimensions, but the footprint of the AD-101 remains constant: 16.51 mm x 43.18 mm.
Terminals for sensor wires
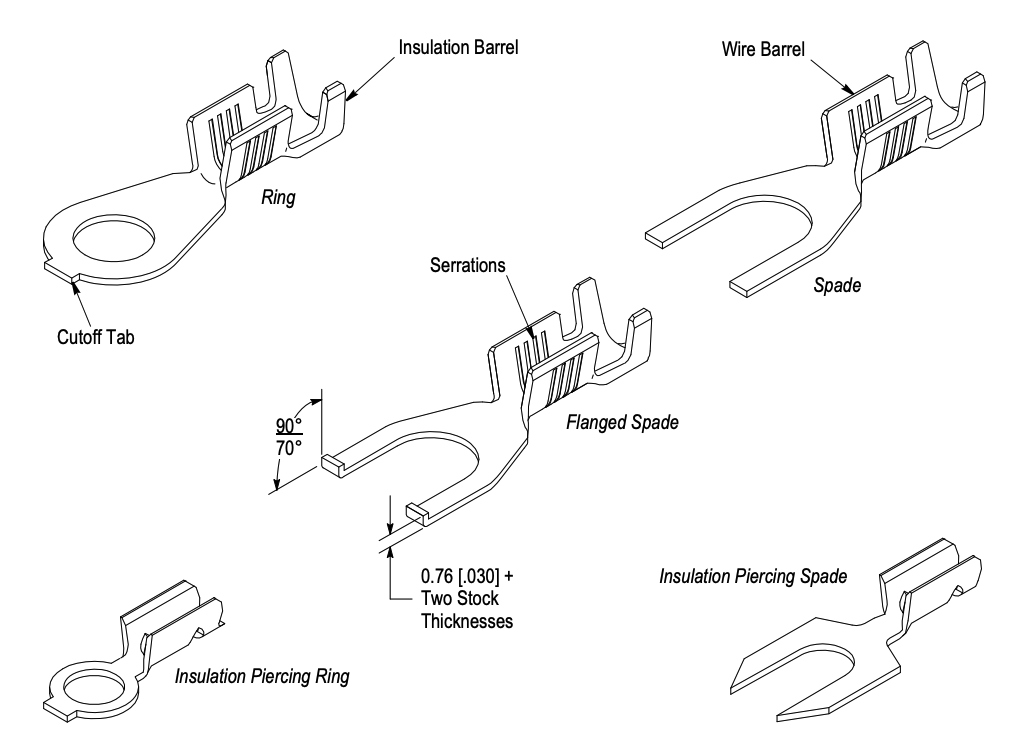
The AD-101 is but one example of a sensor for personal healthcare, there are many others, and they invariably need a terminal to form electrical connections to a system. Terminal options vary as much, if not more, than the types of sensors (Figure 3). However, for designers of personal healthcare subsystems, robust and electrically pure connections with high pull tolerance are important parameters, as are multiple coating options and jacket color-coding capability. Having one supplier that can meet all these sensor connection requirements through off-the-shelf standard components or through custom solutions, can greatly simplify the selection and design-in process.

In the case of the AD-101 sensor, for example, its 28 AWG cables can be matched to the AMPLIVAR ring 22 to 29 AWG 10 TPBR terminal (Figure 2, again). This has a ring tongue with a size 10 stud.
The ring tongue is advantageous in that it helps ensure a physically strong connection, but it requires the screw to be completely removed in order to remove the terminal. In situations where the sensor may need to be detached intermittently, there are various spade options (Figure 4).
Advanced concepts in connectors
In many instances spade or ring terminals can provide placement flexibility for sensor wiring, but with as the number of sensors increases, tidy harnesses with connectors to boards or to other cables may be a preferred option. There are many connector options, but designers need to look first for stability, reliability, and durability. Some connectors provide both physical and audible “snap” features to ensure it has been placed correctly.
For example, the GRACE INERTIATM connectors with 2.5 mm pitch have an inertia locking mechanism that prevents defective products resulting from half-mated connections (Figure 5). The mechanism also prevents connectors from being disconnected due to movement or transportation, both of which are usage patterns in personal healthcare monitoring systems and devices.

The GRACE INERTIAtm connectors come with four keying methods and colors, are rated at 3 A (max), 50 VAC/VDC, and meets GWT requirement per IEC 60335-1, 5th edition (750˚C, without flame), and UL 94 V-0 flammability.
Conclusion
Signal integrity from sensors to the central processor is critical if designers are to create systems capable of executing upon the promise of personal healthcare. However, as shown, achieving optimal signal integrity and system reliability requires close attention to every stage of the signal path, as well as an appreciation of the value of a component partner that can provide insight at every level to optimize the system and prevent re-designs. However, it’s important to connect with a suitable supplier early in the design process, rather than later, to take full advantage of their market and design expertise.
With its position at the heart of enabling sensification in personal healthcare devices, TE Connectivity is one such supplier. It has a broad product portfolio that includes the connectors, sensors, and 5G-ready solutions needed to take personal healthcare devices to the next generation of innovation. It has also demonstrated a track record of developing world-class partnerships to ensure its customers are always close to the solutions they need. One such partnership is with Heilind Electronics, a distributor of electronic components that was built on a foundation of deep inventory, flexible policies, responsiveness, and a commitment to customer service and satisfaction.
From blood pressure sensing to monitoring devices, TE Connectivity and Heilind Electronics ensure designers get the sensors and product solutions they need for the collection of localized data to better serve the personal healthcare space.
