With biosensing analog front end (AFE) ICs available from several major suppliers, researchers have a variety of options to investigate more sophisticated health measurement and monitoring techniques for healthcare professionals as well as users. Some of the applications currently under active investigation include identifying acute tissue injury and skeletal muscle fatigue, assessing neuromuscular disorders, tracking fluid shifts during dialysis, assessing joint health, cuffless blood pressure monitoring and respiratory monitoring.
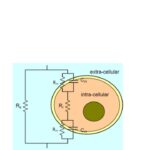
According to PharmiWeb.com report, “Bioimpedance Devices Market is Projected to Reach Total Valuation of US$ 715.1 Million by 2028,” increasing cases of chronic kidney disease (CKD) are expected to contribute to the growth of the bioimpedance devices market. The glomerular filtration rate (GFR) is the best overall index for studying kidney function. Non-invasive BIA is used for improved estimation of GFR in non-diabetic patients with CKD.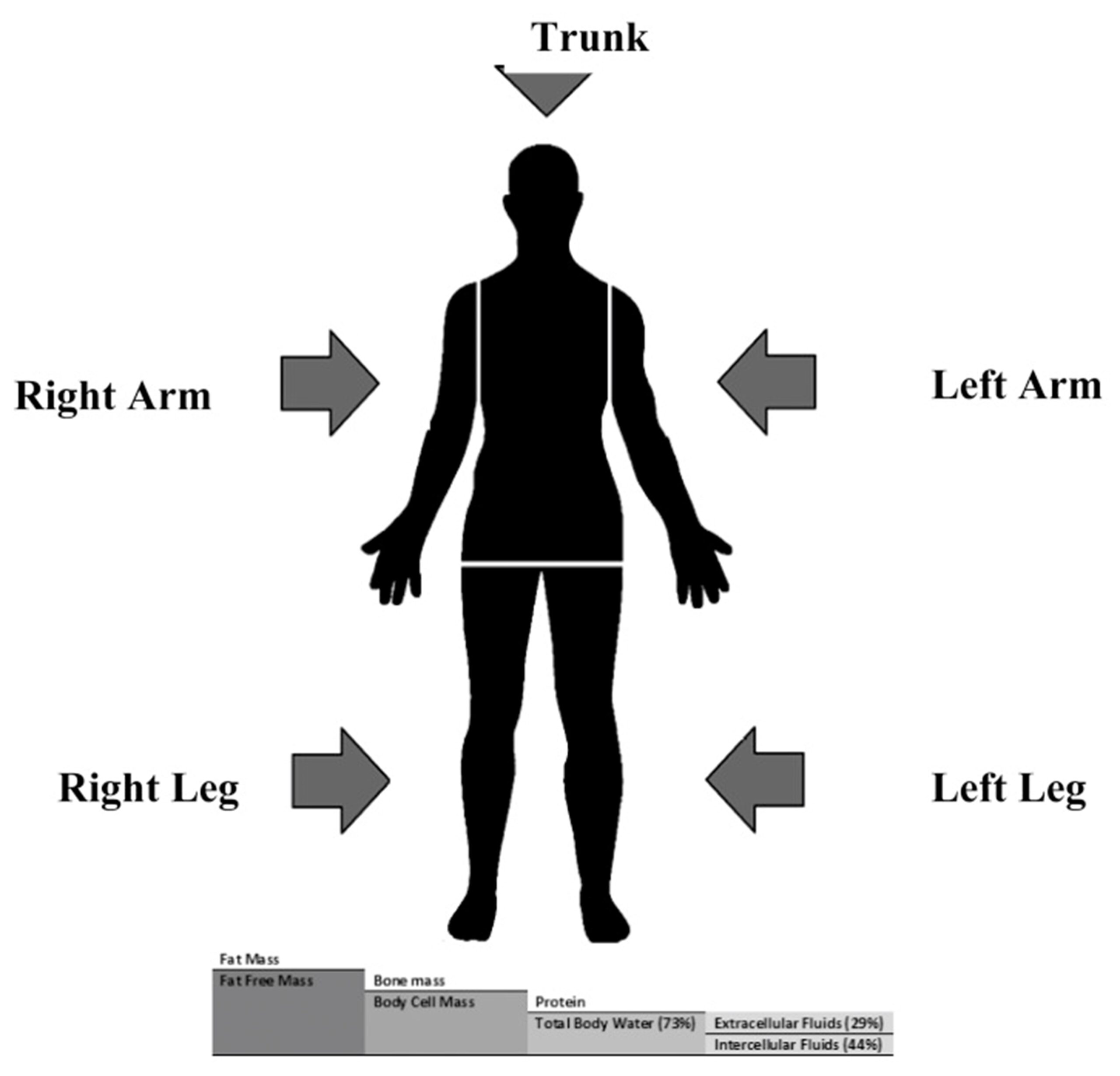
Image source: The Theory and Fundamentals of Bioimpedance Analysis in Clinical Status Monitoring and Diagnosis of Diseases – PMC (nih.gov)
Addressing obesity is another key use of BIA. Upper body (hand-to-hand) BIA is useful for estimating visceral and abdominal fat and small form-factor wearable devices such as a wristwatch can provide this data. In contrast, lower body (leg-to-leg) BIA is useful for estimation of subcutaneous fat and is often obtained from a scale while acquiring weight information.
Different frequencies and multiple frequencies are among BIA aspects that are being explored for and implemented in clinical class products. Measurements at very low frequencies (in the range 1 to 5 kHz) have issues with cell membranes. In contrast, as the frequency increases, the phase angle and capacitive resistance (reactance) increase up to a maximum frequency of about 50 kHz. The use of multi-frequency analysis provides an improved differentiation for some metrics.
ImpediMed pioneered the use of bioimpedance spectroscopy (BIS) technology, one type of BIA, and produced the first commercially available BIS devices in 1990. Their patented technology measures impedance at 256 different frequencies, from 3 kHz to 1000 kHz and uses validated mathematical models to determine three pure resistance values in the body.
As noted in part 1, electrodes are one of the key portions of BIA measurements. The electrodes consist of two current-driving and two voltage-sensing electrodes. Most commercial BIA devices utilize large (4 × 1400 mm2) electrodes to counteract the effect of contact resistance that can cause measurement errors. Researchers have compensated for contact resistance by performing 4-point and 2-point measurements using 68 mm2 outer electrodes and 128 mm2 inner electrodes.
References for further reading:
[1] https://www.mdpi.com/1424-8220/21/9/3013/pdf
[3] https://www.bioscan.com/dtr_bioscan_bia.html
[4] https://www.impedimed.com/resources/bis-technology/
[5] https://www.nature.com/articles/s41598-020-79667-3