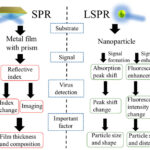
Part 1 of this blog explored research into localized surface plasmon resonance (LSPR) sensors. This part discusses a surface plasmon resonance/surface-enhanced Raman scattering (SPR/SERS) dual-mode plasmonic biosensor and two other sensing techniques.
The SPR/SERS biosensor based on catalytic hairpin assembly (CHA)-induced Au nanoparticle (AuNP) network was proposed for highly sensitive and reliable detection of a short non-coding ribonucleic acid (RNA), a cancer-related microRNA (miRNA) called miRNA-652. A RNA’s principal role is to carry instructions from deoxyribonucleic acid (DNA) for controlling the synthesis of proteins.
Using two probes, networks were formed via the target-triggered CHA reactions. This resulted in the color change of dark-field microscopy (DFM) images and enhanced SERS effect. The SPR sensing was achieved from the integral optical density of dark-field color in DFM images and the SERS sensing was realized by ratiometric SERS signals. With the complementary advantages of the two modes, the combined SPR/SERS dual-mode sensor can provide more options for early detection and double checking the results for cancer-related nucleic acids detection in early disease stage.
Measuring the refractive index of tissues is one of the hot topic areas in bio-optics. Researchers have reported two optical biosensor techniques for detecting cancer cells. The structures consist of one-dimensional photonic crystal (PC) lattices coupled to two metal–insulator–metal (MIM) plasmonic waveguides. Similar to light that consists of photons, plasma oscillations consist of plasmons. Surface plasmon polaritons (SPPs) are the electromagnetic surface waves that propagate at the surface of metal and insulator materials. SPPs can be used to overcome the diffraction limit and manipulate light at a sub-wavelength scale.
In the second topology, a tapering method is used to improve the matching between the MIM plasmonic waveguides and PC structure. The PC lattices at the central part of the structures generate photonic bandgaps (PBGs) with sharp edges in the transmission spectra of the biosensors. In addition to the sharp edges that makes the techniques suitable candidates for sensing applications there is excellent separation between two PBG edges. When the low PBG edge is used as a sensing mechanism, it does not overlap with the high PBG edge by changing the refractive index of the analyte. Consequently, the proposed biosensors can be used for a wide wavelength range.
In addition to being a source of energy for daily activities of the human body, glucose is regarded as a clinical biomarker. An abnormal glucose level in the blood can lead to many endocrine metabolic diseases, including diabetes, hyperglycemia and other diseases. Current sensing methods mainly depend on natural enzymes. These enzymes are unstable, hard to prepare, and expensive, which limits the applications in clinics. Instead, simple, accurate, and sensitive methods for glucose detection are required since glucose level in cancer cells is higher than that in healthy cells. A dual-mode Cu2O nanoparticle (NP) based biosensor for glucose analysis based on colorimetric assay and laser desorption/ionization mass spectrometry (LDI MS) may provide the answer. The investigated method achieved visual and accurate quantitative analysis of glucose in serum and possesses promising application values in clinical disease diagnostics and monitoring.
References
Frontiers | A Copper-Based Biosensor for Dual-Mode Glucose Detection (frontiersin.org)